Description
4SC-202, CAS No. 1186222-89-8, is a potent, selective and orally bioavailable HDAC/LSD1 dual Inhibitor. It inhibits class I HDAC with IC50 of 1.20µM, 1.12 µM, and 0.57µM for HDAC1, HDAC2, and HDAC3, respectively. It also displays inhibitory activity against Lysine specific demethylase 1 (LSD1). 4SC-202 has very high selectivity over ClassIIa/IIb/III HDACs. In HeLa cells, it induces hyperacetylation of histone H3 with EC50 of 1.1 µM. It induces a G2/M cell cycle arrest by interfering with the normal development of the mitotic spindle and causing collapsed spindle apparatus and multiple nucleation centres. In addition, 4SC-202 shows a broad anti-proliferative activity towards human cancer cell lines with a mean IC50 of 0.7 µM. In vivo it shows pronounced and robust anti-tumor activity in both A549 NSCLC xenograft and RKO27 colon carcinoma model. Currently it is in phase I trials for patients with advanced haematological tumours.
Product information
CAS Number: 1186222-89-8
Molecular Weight: 619.71
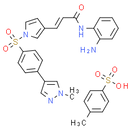
Formula: C30H29N5O6S2
Synonym:
domatinostat tosylate
4SC-202 tosylate
Domatinostat (tosylate)
(E)-N-(2-aminophenyl)-3-(1-((4-(1-methyl-1H-pyrazol-4-yl)phenyl)sulfonyl)-1H-pyrrol-3-yl)acrylamide
(E)-N-(2-aminophenyl)-3-(1-((4-(1-methyl-1H-pyrazol-4-yl)phenyl)sulfonyl)-1H-pyrrol-3-yl)acrylamide 4-methylbenzenesulfonate
Related CAS Number:
1186222-89-8
Chemical Name: (E)-N-(2-aminophenyl)-3-(1-((4-(1-methyl-1H-pyrazol-4-yl)phenyl)sulfonyl)-1H-pyrrol-3-yl)acrylamide tosylic acid
Smiles: CN1C=C(C=N1)C1C=CC(=CC=1)S(=O)(=O)N1C=C(/C=C/C(=O)NC2=CC=CC=C2N)C=C1.CC1C=CC(=CC=1)S(O)(=O)=O
InChiKey: IAVXAZDVNICKFJ-ICSBZGNSSA-N
InChi: InChI=1S/C23H21N5O3S.C7H8O3S/c1-27-16-19(14-25-27)18-7-9-20(10-8-18)32(30,31)28-13-12-17(15-28)6-11-23(29)26-22-5-3-2-4-21(22)24;1-6-2-4-7(5-3-6)11(8,9)10/h2-16H,24H2,1H3,(H,26,29);2-5H,1H3,(H,8,9,10)/b11-6+;
Technical Data
Appearance: Solid powder
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: DMSO up to 50 mM
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical. This product is stable enough for a few weeks during ordinary shipping and time spent in Customs.
Storage Condition: Powder: 4oC for 1 year.
Shelf Life: ≥60 months if stored properly.
Stock Solution Storage: DMSO: 4oC for 3 months.
Drug Formulation: This drug may be formulated in DMSO
HS Tariff Code: 2934.99.9001
How to use
In Vitro:
4SC-202 was used at 10µM final concentration in various in vitro assays.
In Vivo:
4SC-202 was dosed to A549 NSCLC xenograft model and RKO27 colon carcinoma xenografts model orally at 120 mg/Kg once per day.
Products are for research use only. Not for human use.
Documents
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.