Description
AG-120 (Ivosidenib), CAS No. 1448347-49-6, is a potent, selective and reversible inhibitor of IDH1 with IC50 < 100 nM. It has been shown to lower 2-HG levels and restore cellular differentiation in IDH1-mutant primary human blast cells cultured ex vivo. In phase I clinical trials targeting patients with IDH1-mutated, relapsed and/or refractory (R/R) AML or untreated AML not eligible for standard of care or R/R MDS, AG-120 is well tolerated to date (10/2015) and MTD was not reached. Overall response rate is 35% including 12 CRs.
Product information
CAS Number: 1448347-49-6
Molecular Weight: 582.96
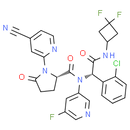
Formula: C28H22ClF3N6O3
Chemical Name: (S)-N-((S)-1-(2-chlorophenyl)-2-((3,3-difluorocyclobutyl)amino)-2-oxoethyl)-1-(4-cyanopyridin-2-yl)-N-(5-fluoropyridin-3-yl)-5-oxopyrrolidine-2-carboxamide
Smiles: N#CC1C=C(N=CC=1)N1[C@@H](CCC1=O)C(=O)N([C@@H](C1=CC=CC=C1Cl)C(=O)NC1CC(F)(F)C1)C1=CC(F)=CN=C1
InChiKey: WIJZXSAJMHAVGX-DHLKQENFSA-N
InChi: InChI=1S/C28H22ClF3N6O3/c29-21-4-2-1-3-20(21)25(26(40)36-18-11-28(31,32)12-18)37(19-10-17(30)14-34-15-19)27(41)22-5-6-24(39)38(22)23-9-16(13-33)7-8-35-23/h1-4,7-10,14-15,18,22,25H,5-6,11-12H2,(H,36,40)/t22-,25-/m0/s1
Technical Data
Appearance: Solid Power.
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: DMSO up to 100 mM; EtOH up to 100 mM
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined.
HS Tariff Code: 382200
How to use
In Vitro:
AG-120 was used at 1 µM in vitro and cellular assays.
In Vivo:
AG-120 was dosed orally to mice bearing IDH1 mutant tumors at 150 mg/Kg once per day.
References:
- Kc B, et al. Evidence for Clinical Differentiation and Differentiation Syndrome in Patients With Acute Myeloid Leukemia and IDH1 Mutations Treated With the Targeted Mutant IDH1 Inhibitor, AG-120. (2016) Clin Lymphoma Myeloma Leuk. In press.
- Erica Hansen, et al. AG-120, an Oral, Selective, First-in-Class, Potent Inhibitor of Mutant IDH1, Reduces Intracellular 2HG and Induces Cellular Differentiation in TF-1 R132H Cells and Primary Human IDH1 Mutant AML Patient Samples Treated Ex Vivo. (2014) Blood. 124:3734
Products are for research use only. Not for human use.
Documents
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.