Description
(-)-Narwedine, CAS No. 510-77-0, is a cholinesterase (AChE) inhibitor for the treatment of mild to moderate Alzheimer's disease.
Product information
CAS Number: 510-77-0
Molecular Weight: 285.34
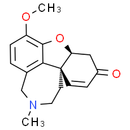
Formula: C17H19NO3
Synonym:
(-)-Narwedine
Galanthaminone
Narwedine
3-Deoxy-3-oxo-galanthamine
Chemical Name: (4aS,8aS)-4a,5,9,10,11,12-Hexahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-one
Smiles: COC1=CC=C2CN(C)CC[C@@]34C=CC(=O)C[C@@H]3OC1=C42
InChiKey: QENVUHCAYXAROT-YOEHRIQHSA-N
InChi: InChI=1S/C17H19NO3/c1-18-8-7-17-6-5-12(19)9-14(17)21-16-13(20-2)4-3-11(10-18)15(16)17/h3-6,14H,7-10H2,1-2H3/t14-,17-/m0/s1
Technical Data
Appearance: Solid Power.
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: Soluble in DMSO
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined.
HS Tariff Code: 382200
How to use
In Vitro:
(-)-Narwedine reduces the action of AChE and therefore tends to increase the concentration of acetylcholine in the brain. is also an allosteric ligand at nicotinic acetylcholine receptors. It has shown activity in modulating the nicotinic cholinergic receptors on cholinergic neurons to increase acetylcholine release.
In Vivo:
Absorption of (-)-Narwedine is rapid and complete and shows linear pharmacokinetics. It is well absorbed with absolute oral bioavailability between 80 and 100%. It has a half-life of seven hours. Peak effect of inhibiting acetylcholinesterase was achieved about one hour after a single oral dose of 8 mg in some healthy volunteers.
References:
- Bickel U, Thomsen T, Weber W, Fischer JP, Bachus R, Nitz M, Kewitz H.Pharmacokinetics of galanthamine in humans and corresponding cholinesterase inhibition. Clin Pharmacol Ther. 1991 Oct;50(4):420-8. PubMed PMID: 1914378.
- Thomsen T, Bickel U, Fischer JP, Kewitz H. Stereoselectivity of cholinesterase inhibition by galanthamine and tolerance in humans. Eur J Clin Pharmacol.1990;39(6):603-5. PubMed PMID: 2095347.
- Mihailova D, Yamboliev I, Zhivkova Z, Tencheva J, Jovovich V. Pharmacokinetics of galanthamine hydrobromide after single subcutaneous and oral dosage in humans. Pharmacology. 1989;39(1):50-8. PubMed PMID: 2587617.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.