Description
ABT-751, also known as E7010, CAS No. 141430-65-1, is an orally bioavailable antimitotic sulfonamide. ABT-751 binds to the colchicine-binding site on beta-tubulin and inhibits the polymerization of microtubules, thereby preventing tumor cell replication. This agent also disrupts tumor neovascularization, reducing tumor blood flow and so inducing a cytotoxic effect.
Product information
CAS Number: 141430-65-1
Molecular Weight: 371.41
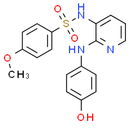
Formula: C18H17N3O4S
Synonym:
ABT751
ABT-751
E7010
ABT 751
E-7010
E 7010
Chemical Name: N-(2-((4-hydroxyphenyl)amino)pyridin-3-yl)-4-methoxybenzenesulfonamide
Smiles: COC1C=CC(=CC=1)S(=O)(=O)NC1=CC=CN=C1NC1C=CC(O)=CC=1
InChiKey: URCVCIZFVQDVPM-UHFFFAOYSA-N
InChi: InChI=1S/C18H17N3O4S/c1-25-15-8-10-16(11-9-15)26(23,24)21-17-3-2-12-19-18(17)20-13-4-6-14(22)7-5-13/h2-12,21-22H,1H3,(H,19,20)
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: Soluble in DMSO, not in water
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined.
HS Tariff Code: 382200
References:
- Gaynon PS, Harned TM; Therapeutic Advances in Childhood Leukemia/Lymphoma Consortium. ABT-751 in relapsed childhood acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 2012 Oct;34(7):583-4. PubMed PMID: 22584778.
- Ma T, Fuld AD, Rigas JR, Hagey AE, Gordon GB, Dmitrovsky E, Dragnev KH. A phase I trial and in vitro studies combining ABT-751 with carboplatin in previously treated non-small cell lung cancer patients. Chemotherapy. 2012;58(4):321-9. doi: 10.1159/000343165. Epub 2012 Nov 12. PubMed PMID: 23147218; PubMed Central PMCID: PMC3570826.
- Fox E, Mosse' YP, Meany HM, Gurney JG, Khanna G, Jackson HA, Gordon G, Shusterman S, Park JR, Cohn SL, Adamson PC, London WB, Maris JM, Balis FM. Time to disease progression in children with relapsed or refractory neuroblastoma treated with ABT-751: a report from the Children's Oncology Group (ANBL0621). Pediatr Blood Cancer. 2014 Jun;61(6):990-6. doi: 10.1002/pbc.24900. Epub 2013 Dec 18. PubMed PMID: 24347462.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.