Description
Afuresertib HCl, CAS No. 1047645-82-8, also known as GSK2110183, is an orally bioavailable inhibitor of the serine/threonine protein kinase Akt (protein kinase B) with potential antineoplastic activity. Akt inhibitor GSK2110183 binds to and inhibits the activity of Akt, which may result in inhibition of the PI3K/Akt signaling pathway and tumor cell proliferation and the induction of tumor cell apoptosis. Activation of the PI3K/Akt signaling pathway is frequently associated with tumorigenesis and dysregulated PI3K/Akt signaling may contribute to tumor resistance to a variety of antineoplastic agents.
Product information
CAS Number: 1047645-82-8
Molecular Weight: 463.78
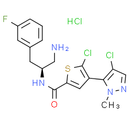
Formula: C18H18Cl3FN4OS
Related CAS Number:
1047634-63-8 (free base)
Chemical Name: N-((S)-1-amino-3-(3-fluorophenyl)propan-2-yl)-5-chloro-4-(4-chloro-1-methyl-1H-pyrazol-5-yl)thiophene-2-carboxamide hydrochloride
Smiles: Cl.CN1N=CC(Cl)=C1C1C=C(SC=1Cl)C(=O)N[C@H](CN)CC1=CC(F)=CC=C1
InChiKey: YFQJOPFTGMHYNV-YDALLXLXSA-N
InChi: InChI=1S/C18H17Cl2FN4OS.ClH/c1-25-16(14(19)9-23-25)13-7-15(27-17(13)20)18(26)24-12(8-22)6-10-3-2-4-11(21)5-10;/h2-5,7,9,12H,6,8,22H2,1H3,(H,24,26);1H/t12-;/m0./s1
Technical Data
Appearance: Solid Power.
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: Soluble in DMSO, not in water
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined.
HS Tariff Code: 382200
How to use
In Vitro:
Afuresertib (GSK 2110183) exhibits favorable tumor-suppressive effects on malignant pleural mesothelioma (MPM) cells. Afuresertib significantly increases caspase-3 and caspase-7 activities and apoptotic cell number among ACC-MESO-4 and MSTO-211H cells. Afuresertib strongly arrests the cell cycle in the G1 phase. Western blotting analysis shows that Afuresertib increases the expression of p21WAF1/CIP1 and decreases the phosphorylation of Akt substrates, including GSK-3β and FOXO family proteins. Afuresertib-induced p21 expression promotes G1 phase arrest by inducing FOXO activity. Afuresertib significantly enhances cisplatin-induced cytotoxicity. Afuresertib modulates the expression E2F1 and MYC, which are associated with fibroblast core serum response.
In Vivo:
Mice bearing BT474 breast tumor xenografts are dosed orally with either vehicle or GSK2110183 at 10, 30 or 100 mg/kg daily for 21 days which result in 8, 37 and 61% TGI, respectively. Mice tolerated GSK2110183 well, with 1-3% body weight loss reported after 5 days of dosing which recover over the course of the study. Other tumor xenograft models which possess an activation of the Akt pathway are explored to further demonstrate compound efficacy. Mice treated with GSK2110183 at 10, 30 and 100 mg/kg result in 23, 37 and 97% TGI, respectively, of SKOV3 xenografts.
References:
- Naymagon L, Abdul-Hay M. Novel agents in the treatment of multiple myeloma: a review about the future. J Hematol Oncol. 2016 Jun 30;9(1):52. doi: 10.1186/s13045-016-0282-1. Review. PubMed PMID: 27363832; PubMed Central PMCID: PMC4929712.
- Yamaji M, Ota A, Wahiduzzaman M, Karnan S, Hyodo T, Konishi H, Tsuzuki S, Hosokawa Y, Haniuda M. Novel ATP-competitive Akt inhibitor afuresertib suppresses the proliferation of malignant pleural mesothelioma cells. Cancer Med. 2017 Nov;6(11):2646-2659. doi: 10.1002/cam4.1179. Epub 2017 Sep 27. PubMed PMID: 28960945; PubMed Central PMCID: PMC5673922.
- Kinoshita S, Ri M, Kanamori T, Aoki S, Yoshida T, Narita T, Totani H, Ito A, Kusumoto S, Ishida T, Komatsu H, Iida S. Potent antitumor effect of combination therapy with sub-optimal doses of Akt inhibitors and pomalidomide plus dexamethasone in multiple myeloma. Oncol Lett. 2018 Jun;15(6):9450-9456. doi: 10.3892/ol.2018.8501. Epub 2018 Apr 16. PubMed PMID: 29928335; PubMed Central PMCID: PMC6004690.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.