Description
Belinostat, HDAC inhibitor, CAS No. 866323-14-0, is a novel hydroxamic acid-type histone deacetylase (HDAC) inhibitor with antineoplastic activity. Belinostat targets HDAC enzymes, thereby inhibiting tumor cell proliferation, inducing apoptosis, promoting cellular differentiation, and inhibiting angiogenesis. This agent may sensitize drug-resistant tumor cells to other antineoplastic agents, possibly through a mechanism involving the down-regulation of thymidylate synthase. Belinostat was approved in 2014 for relapsed or refractory peripheral T-cell lymphoma.
Product information
CAS Number: 866323-14-0
Molecular Weight: 318.35
Formula: C15H14N2O4S
Synonym:
PXD101
PX105684
PXD-101
PXD 101
PX-105684
PX 105684
NSC726630
Beleodaq
Related CAS Number:
866323-14-0 (Belinostat(E))
414864-00-9 (Belinostat(EZ))
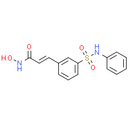
Chemical Name: (E)-N-hydroxy-3-[3-(phenylsulfamoyl)phenyl]prop-2-enamide.
Smiles: ONC(=O)/C=C/C1=CC(=CC=C1)S(=O)(=O)NC1C=CC=CC=1
InChiKey: NCNRHFGMJRPRSK-MDZDMXLPSA-N
InChi: InChI=1S/C15H14N2O4S/c18-15(16-19)10-9-12-5-4-8-14(11-12)22(20,21)17-13-6-2-1-3-7-13/h1-11,17,19H,(H,16,18)/b10-9+
Technical Data
Appearance: Solid Power.
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: Soluble in DMSO, not in water
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined.
HS Tariff Code: 382200
How to use
In Vitro:
Belinostat (PXD101) induces a concentration-dependent (0.2-5 μM) increase in acetylation of histone H4 in tumor cell lines. Belinostat is cytotoxic in vitro in a number of tumor cell lines with IC50s in the range 0.2-3.4 μM as determined by a clonogenic assay and induces apoptosis. Belinostat inhibits the growth of a number of human tumor cell lines in vitro with IC50s determined by a clonogenic assay in the range 0.2-3.4 μM. Belinostat (PXD101) is a potent histone deacetylase (HDAC) inhibitor, potently inhibits the enzymatic activity of purified recombinant HDAC6 (IC50 of 82 nM).
In Vivo:
Treatment of nude mice bearing human ovarian and colon tumor xenografts with Belinostat (10-40 mg/kg/day i.p.) daily for 7 days causes a significant dose-dependent growth delay with no obvious signs of toxicity to the mice. Growth delay is also observed for xenografts of cisplatin-resistant ovarian tumor cells. A marked increase in acetylation of H4 is detected in blood and tumor of mice 3 h after treatment with Belinostat (PXD101). The inhibition of growth of human tumor xenografts in mice, with no apparent toxicity. Belinostat (PXD101) displays single-agent antitumor activity on human A2780 ovarian cancer s.c. xenografts which is enhanced via combination therapy with Carboplatin.
References:
- Odenike O, Halpern A, Godley LA, Madzo J, Karrison T, Green M, Fulton N, Mattison RJ, Yee KW, Bennett M, Koval G, Malnassy G, Larson RA, Ratain MJ, Stock W. A phase I and pharmacodynamic study of the histone deacetylase inhibitor belinostat plus azacitidine in advanced myeloid neoplasia. Invest New Drugs. 2014 Dec 9. [Epub ahead of print] PubMed PMID: 25483416.
- Kim MJ, Lee JS, Park SE, Yi HJ, Jeong IG, Kang JS, Yun J, Lee JY, Ro S, Lee JS, Choi EK, Hwang JJ, Kim CS. Combination treatment of renal cell carcinoma with belinostat and 5-fluorouracil: A role for oxidative stress-induced DNA damage and HSP90-regulated thymidine synthase. J Urol. 2014 Nov 26. pii: S0022-5347(14)05012-5. doi: 10.1016/j.juro.2014.11.091. [Epub ahead of print] PubMed PMID: 25433307.
- Foss F, Advani R, Duvic M, Hymes KB, Intragumtornchai T, Lekhakula A, Shpilberg O, Lerner A, Belt RJ, Jacobsen ED, Laurent G, Ben-Yehuda D, Beylot-Barry M, Hillen U, Knoblauch P, Bhat G, Chawla S, Allen LF, Pohlman B. A Phase II trial of Belinostat (PXD101) in patients with relapsed or refractory peripheral or cutaneous T-cell lymphoma. Br J Haematol. 2014 Nov 17. doi: 10.1111/bjh.13222. [Epub ahead of print] PubMed PMID: 25404094.
- Bodiford A, Bodge M, Talbott MS, Reddy NM. Profile of belinostat for the treatment of relapsed or refractory peripheral T-cell lymphoma. Onco Targets Ther. 2014 Oct 24;7:1971-7. doi: 10.2147/OTT.S59269. eCollection 2014. Review. PubMed PMID: 25368524; PubMed Central PMCID: PMC4216035.
- Thomas A, Rajan A, Szabo E, Tomita Y, Carter CA, Scepura B, Lopez-Chavez A, Lee MJ, Redon CE, Frosch A, Peer CJ, Chen Y, Piekarz R, Steinberg SM, Trepel JB, Figg WD, Schrump DS, Giaccone G. A phase I/II trial of belinostat in combination with cisplatin, doxorubicin, and cyclophosphamide in thymic epithelial tumors: a clinical and translational study. Clin Cancer Res. 2014 Nov 1;20(21):5392-402. doi: 10.1158/1078-0432.CCR-14-0968. Epub 2014 Sep 4. PubMed PMID: 25189481; PubMed Central PMCID: PMC4216756.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.