Description
Suprofen is a COX12 inhibitor and a non-steroidal anti-inflammatory drug (NSAID).
Product information
CAS Number: 40828-46-4
Molecular Weight: 260.31
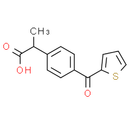
Formula: C14H12O3S
Synonym:
TN-762
R-25061
Suprofenum
Profenal
Chemical Name: 2-(4-(thiophene-2-carbonyl)phenyl)propanoic acid
Smiles: CC(C(O)=O)C1C=CC(=CC=1)C(=O)C1=CC=CS1
InChiKey: MDKGKXOCJGEUJW-UHFFFAOYSA-N
InChi: InChI=1S/C14H12O3S/c1-9(14(16)17)10-4-6-11(7-5-10)13(15)12-3-2-8-18-12/h2-9H,1H3,(H,16,17)
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: DMSO: 52 mg/mL(199.76 mM). Water: Insoluble.
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined
HS Tariff Code: 382200
How to use
In Vitro:
Suprofen inactivates the diclofenac-4-hydroxylase activity of baculovirus-expressed P450 2C9 in a time- and concentration-dependent manner, which is consistent with mechanism-based inactivation. [1] Suprofen is a known generator of singlet oxygen whose participation in the reaction is supported by the effects of quenchers and scavengers. Suprofen photosensitizes the production of alkali-labile cleavage sites in DNA much more efficiently than direct strand breaks. [2] Suprofen sensitizes photo-oxidation of cholesterol, producing 7 alpha- and 7 beta-hydroperoxides but not 5 alpha-hydroperoxide of cholesterol. Suprofen-sensitized photo-oxidation of membrane lipids of liposomes increases leakage of trapped glucose, suggesting photosensitized destabilization of the membrane structure. [3] Suprofen incorporation in the phosphatidylcholine (PC) evaporation vesicles (REV) membrane leads to approximately 5% increase in the encapsulation efficiency (34%) in comparison to standard REV (29%). [4]
In Vivo:
Suprofen combined with PGF2 alpha blocks induction of uterine contractions, suggesting the possibility that Suprofen also antagonizes PGF2 alpha receptor binding. [5] Suprofen are effective at preventing BAB disruption after paracentesis in dogs, indicating their potential usefulness for treatment of prostaglandin-mediated ocular disease. [6] Suprofen (3.7 mg/kg, i.v.) induces a marked decrease in the firing evoked in arthritic rats by ankle mobilization. [7]
References:
- Yeadon, A., et al., Suprofen. An overview of long-term safety. Pharmacology, 1983. 27 Suppl 1: p. 87-94.
- Dawood, M.Y. and F.S. Khan-Dawood, Differential suppression of menstrual fluid prostaglandin F2a, prostaglandin E2, 6-keto prostaglandin F1a and thromboxane B2 by suprofen in women with primary dysmenorrhea. Prostaglandins Other Lipid Mediat, 2007. 83(1-2): p. 146-53.
- Fujita H, et al. Photodermatol Photoimmunol Photomed, 1992-1993, 9(5), 203-208.
- Starrs SM, et al. Photochem Photobiol, 2000, 72(3), 291-297.
- O'Donnell JP, et al. Drug Metab Dispos, 2003, 31(11), 1369-1377.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.