Description
Sitagliptin, previously identified as MK-0431 and marketed as the phosphate salt under the trade name Januvia) is an oral antihyperglycemic (antidiabetic drug) of the dipeptidyl peptidase-4 (DPP-4) inhibitor class.
Product information
CAS Number: 486460-32-6
Molecular Weight: 407.31
Formula: C16H15F6N5O
Synonym:
EC 690-730-1
HSDB 7516
HSDB7516
HSDB-7516
Januvia
LEZ 763
LEZ-763
LEZ763
Tesavel
Xelevia
Related CAS Number:
654671-77-9 (Sitagliptin Phosphate Monohydrate)
654671-78-0 (Sitagliptin Phosphate)
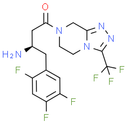
Chemical Name: (R)-3-amino-1-(3-(trifluoromethyl)-5, 6-dihydro-[1, 2, 4]triazolo[4, 3-a]pyrazin-7(8H)-yl)-4-(2, 4, 5-trifluorophenyl)butan-1-one
Smiles: N[C@H](CC1=CC(F)=C(F)C=C1F)CC(=O)N1CC2=NN=C(N2CC1)C(F)(F)F
InChiKey: MFFMDFFZMYYVKS-SECBINFHSA-N
InChi: InChI=1S/C16H15F6N5O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22/h4,6,9H,1-3,5,7,23H2/t9-/m1/s1
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: DMSO: 81 mg/mL(198.86 mM). Water: undefined.
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined
HS Tariff Code: 382200
How to use
In Vitro:
Sitagliptin exhibits a > 2600-fold margin of selectivity against DPP8, DPP9, and other members of the dipeptidyl peptidase family (i.e., potency against DPP-4 vs. DPP8/9). MK0431 reduces in vitro migration of isolated splenic CD4 T-cells through a pathway involving cAMP/PKA/Rac1 activation. Sitagliptin exerts a novel, direct action in order to stimulate GLP-1 secretion by the intestinal L cell through a DPP-4-independent, protein kinase A- and MEK-ERK1/2-dependent pathway. It therefore reduces the effect of autoimmunity on graft survival.
In Vivo:
Sitagliptin is well absorbed after oral administration with a bioavailability of 87%. Sitagliptin has an apparent terminal half-life of 10–12 h at doses of 25-100 mg and is excreted mainly (≈ 80%) as unchanged compound by the kidneys. Sitagliptin does not interfere with the P450 cytochrome enzymes nor have there been any reported significant drug-drug interactions. Sitagliptin has been shown to inhibit DPP-4 activity by > 90% within 1-2 h of administration. It has a short half-life in mice (1-2 h). Chronic sitagliptin treatment in a non-geneticmouse model of type 2 diabetes elicits significant improvement in glycemic control. The improved glucose homeostasis correlates with restoration of normal islet cell (α and β cells) mass, architecture and insulin secretion capacity in response to glucose stimulation. Sitagliptin prolongs islet graft survival in streptozotocin-induced and NOD mice. Administration of sitagliptin in vivo reduces lymph node and splenic CD4+ T-cell migration, measured in vitro, via incretin- and nonincretin-mediated effects, respectively, and splenic sDPP-IV-responsive CD4+ T-cells and lymph node incretin nonresponsive CD4+ T-cells selectively infiltrated islets of diabetic NOD mice, after tail vein injection. Sitagliptin significantly suppressed epileptogenesis in PTZ (pentylenetetrazole)-induced seizures. Sitagliptin counteracted neuronal damage and all biochemical, and histo-chemical alteration induced by PTZ. Oral sitagliptin can promote hippocampal neurogenesis, counteract hippocampal oxidative stress, and prevent the decline in mice cognition.
References:
- Sangle GV et al. Endocrinology. 2012; 153(2): 564-573.
- Kim SJ et al. Diabetes. 2009; 58(3): 641-651.
- Karasik A, et al. Curr Med Res Opin. 2008, 24(2):489-96.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.