Description
Danoprevir, also known as ITMN-191 and RG-7227, is under development by InterMune Inc and Roche Holding AG, is a promising, potent NS3/4A protease inhibitor for the oral treatment of HCV infection. Preclinical data demonstrated that danoprevir binds with high affinity and dissociates slowly from the HCV NS3 protease, allowing high liver drug exposure with only modest plasma drug exposure. A phase Ib, 'IFN-free' clinical trial demonstrated that danoprevir, combined with the HCV polymerase inhibitor RG-7128 (Pharmasset Inc/Roche Holding AG), was effective in reducing HCV-RNA levels in a large proportion of treatment-naïve patients with HCV infection and in approximately half of previously non-responsive patients with HCV-1 infection, without resistance or safety concerns.
Product information
CAS Number: 850876-88-9
Molecular Weight: 731.83
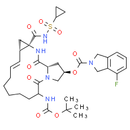
Formula: C35H46FN5O9S
Synonym:
RG7227
RG 7227
RG-7227
ITMN191
ITMN-191
ITMN 191
RO5190591
RO-5190591
RO 5190591
Chemical Name: (2R, 13aS, 14aR, 16aS, Z)-6-((tert-butoxycarbonyl)amino)-14a-((cyclopropylsulfonyl)carbamoyl)-5, 16-dioxo-1, 2, 3, 5, 6, 7, 8, 9, 10, 11, 13a, 14, 14a, 15, 16, 16a-hexadecahydrocyclopropa[e]pyrrolo[1, 2-a][1, 4]diazacyclopentadecin-2-yl 4-fluoroisoindoline-2-carboxylate
Smiles: CC(C)(C)OC(=O)NC1CCCCCC=C[C@@H]2C[C@]2(NC(=O)[C@@H]2C[C@H](CN2C1=O)OC(=O)N1CC2=C(C1)C=CC=C2F)C(=O)NS(=O)(=O)C1CC1 |c:14|
InChiKey: ZVTDLPBHTSMEJZ-QIFYYAQYSA-N
InChi: InChI=1S/C35H46FN5O9S/c1-34(2,3)50-32(45)37-27-13-8-6-4-5-7-11-22-17-35(22,31(44)39-51(47,48)24-14-15-24)38-29(42)28-16-23(19-41(28)30(27)43)49-33(46)40-18-21-10-9-12-26(36)25(21)20-40/h7,9-12,22-24,27-28H,4-6,8,13-20H2,1-3H3,(H,37,45)(H,38,42)(H,39,44)/b11-7-/t22-,23-,27?,28+,35-/m1/s1
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: DMSO: 144 mg/mL(196.76 mM). Water: Insoluble.
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined
HS Tariff Code: 382200
How to use
In Vitro:
Danoprevir (0.29 nM) inhibits the reference genotype 1 NS3/4A protease half-maximally, but a high dose of Danoprevir (10 μM) shows no appreciably inhibition in a panel of 79 proteases, ion channels, transporters, and cell surface receptors. Danoprevir remains bound to and inhibits NS3/4A for more than 5 hours after its initial association. Danoprevir (45 nM) eliminates a patient-derived HCV genotype 1b replicon from hepatocyte-derived Huh7 cells with an EC50 of 1.8 nM. In HCV subgenomic replicon cell lines containing the individual mutations, V36M, R109K, and V170A substitutions confer little or no resistance to Danoprevir, but the R155K substitution confers a high level (62-fold increase) of resistance to Danoprevir. In Huh7.5 cells transfected with chimeric recombinant virus, Danoprevir shows antiviral inhibition effects against HCV genotypes 1, 4 and 6 with IC50 of 2-3 nM, which are >100-fold lower than genotypes 2/3/5 (280-750 nM).
In Vivo:
Danoprevir (30 mg/kg) administered to rats or monkeys shows that its concentrations in liver 12 hours after dosing exceed the Danoprevir concentration required to eliminate replicon RNA from cells.
References:
- Seiwert SD, et al. Antimicrob Agents Chemother, 2008, 52(12), 4432-4441.
- Bartels DJ, et al. J Infect Dis, 2008, 198(6), 800-807.
- Imhof I, et al. Hepatology, 2011, 53(4), 1090-1099.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.