Description
Rivaroxaban, also known as BAY 59-7939, is an oral anticoagulant invented and manufactured by Bayer; in a number of countries it is marketed as Xarelto. If approved by the United States FDA, it will be marketed by Ortho-McNeil Pharmaceutical. It is the first available orally active direct factor Xa inhibitor. Rivaroxaban is well absorbed from the gut and maximum inhibition of factor Xa occurs four hours after a dose. The effects lasts 8–12 hours, but factor Xa activity does not return to normal within 24 hours so once-daily dosing is possible.
Product information
CAS Number: 366789-02-8
Molecular Weight: 435.88
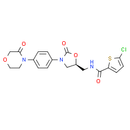
Formula: C19H18ClN3O5S
Synonym:
BAY-59-7939
Chemical Name: (S)-5-chloro-N-((2-oxo-3-(4-(3-oxomorpholino)phenyl)oxazolidin-5-yl)methyl)thiophene-2-carboxamide
Smiles: O=C1COCCN1C1C=CC(=CC=1)N1C[C@H](CNC(=O)C2=CC=C(Cl)S2)OC1=O
InChiKey: KGFYHTZWPPHNLQ-AWEZNQCLSA-N
InChi: InChI=1S/C19H18ClN3O5S/c20-16-6-5-15(29-16)18(25)21-9-14-10-23(19(26)28-14)13-3-1-12(2-4-13)22-7-8-27-11-17(22)24/h1-6,14H,7-11H2,(H,21,25)/t14-/m0/s1
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Solubility: Solubility (25°C) DMSO: 87 mg/mL(199.59 mM). Water: Insoluble.
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined
HS Tariff Code: 382200
How to use
In Vitro:
Rivaroxaban is an oral, direct inhibitor of Factor Xa (FXa), being developed for the prevention and treatment of arterial and venous thrombosis with a Ki of 0.4 nM. Rivaroxaban also inhibits prothrombinase activity with IC50 of 2.1 nM. Rivaroxaban also shows a similar affinity to purified human and rabbit FXa (IC50 0.7 nM and 0.8 nM, respectively), but a lesser potency against purified rat FXa (IC50 3.4 nM). Endogenous human and rabbit FXa in plasma is inhibited to a similar extent by Rivaroxaban (IC50 21 nM and 21 nM, respectively), while 14-fold higher concentrations are required in rat plasma (IC50 290 nM). Rivaroxaban exhibits high permeability and polarized transport across Caco-2 cells as a substrate of the P-gp, but exhibits no inhibitory effect on P-gp-mediated drug transport up to concentrations of 100 μM in vitro.
In Vivo:
Rivaroxaban reduces venous thrombosis in a dose dependent manner (ED50 0.1 mg/kg i.v.) in a rat venous stasis model. Rivaroxaban reduces arterial thrombus formation in an arteriovenous (AV) shunt in rats (ED50 5.0 mg/kg p.o.) and rabbits (ED50 0.6 mg/kg p.o.). Plasma pharmacokinetics of Rivaroxaban are linear across the investigated dose range (1-10 mg/kg in rats, 0.3-3 mg/kg in dogs). Plasma clearance is low: 0.4 L/kg/h in rats and 0.3 L/kg/h in dogs; the volume of distribution (V(ss)) is moderate: 0.3 L/kg in rats, and 0.4 L/kg in dogs. The elimination half-life after oral administration is short in both species (0.9-2.3 hours).
References:
- Weinz C, et al. Xenobiotica, 2005, 35(9), 891-910.
- Gnoth MJ, et al. J Pharmacol Exp Ther, 2011, 338(1), 372-380.
- Perzborn E, et al. J Thromb Haemost, 2005, 3(3), 514–521.
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.