Description
Sitagliptin (INN; previously identified as MK-0431 and sold under the trade name Januvia) is an oral antihyperglycemic (anti-diabetic drug) of the dipeptidyl peptidase-4 (DPP-4) inhibitor class. It was developed and is marketed by Merck & Co. This enzyme-inhibiting drug is used either alone or in combination with other oral antihyperglycemic agents (such as metformin or a thiazolidinedione) for treatment of diabetes mellitus type 2. The benefit of this medicine is its lower side-effects (e.g., less hypoglycemia, less weight gain) in the control of blood glucose values. Exenatide (Byetta) also works by its effect on the incretin system.
Product information
CAS Number: 654671-77-9
Molecular Weight: 523.32
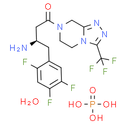
Formula: C16H20F6N5O6P
Synonym:
MK-0431 phosphate monohydrate
Januvia phosphate monohydrate
Chemical Name: (3R)-3-amino-1-[3-(trifluoromethyl)-5H,6H,7H,8H-[1,2,4]triazolo[4,3-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)butan-1-one phosphoric acid hydrate
Smiles: O.N[C@@H](CC(=O)N1CC2=NN=C(N2CC1)C(F)(F)F)CC1=CC(F)=C(F)C=C1F.OP(O)(O)=O
InChiKey: GQPYTJVDPQTBQC-KLQYNRQASA-N
InChi: InChI=1S/C16H15F6N5O.H3O4P.H2O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22;1-5(2,3)4;/h4,6,9H,1-3,5,7,23H2;(H3,1,2,3,4);1H2/t9-;;/m1../s1
Technical Data
Appearance: Solid Power
Purity: ≥98% (or refer to the Certificate of Analysis)
Shipping Condition: Shipped under ambient temperature as non-hazardous chemical or refer to Certificate of Analysis
Storage Condition: Dry, dark and -20 oC for 1 year or refer to the Certificate of Analysis.
Shelf Life: ≥12 months if stored properly.
Stock Solution Storage: 0 - 4 oC for 1 month or refer to the Certificate of Analysis.
Drug Formulation: To be determined
HS Tariff Code: 382200
Products are for research use only. Not for human use.
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.